THE CHEMISTRY BEHIND THE DEPLETION OF THE OZONE LAYER

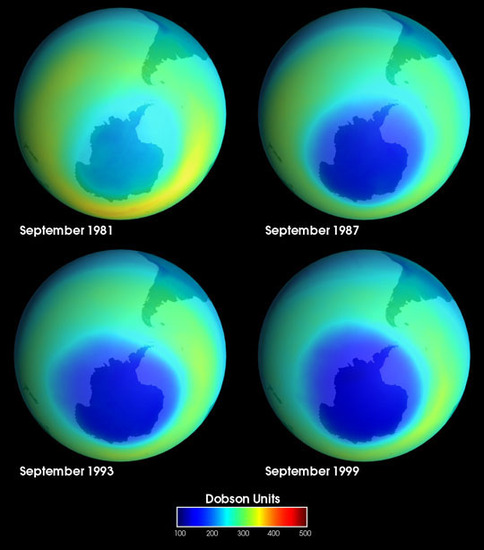
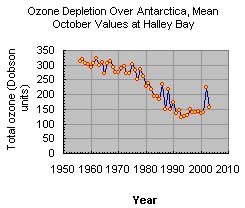
The Earth's ozone layer play an important role in absorbing harmful ultraviolet radiation given off by the sun. In the last century, the ozone layer has been depleted quickly due to chemicals and gases introduced to the atmosphere by humans. There are numerous chemical reactions that have the ability to deplete the stratospheric ozone. Some of these reactions that destroy the ozone involve halogens, such as chlorine and bromine. Ozone, molecular formula O3, is a pale blue gas with a distinct smell.
In the troposphere, or the lowest atmosphere, ozone can produce photochemical smog, which contains major chemical pollutants that pose a threat to the environment. It is a powerful oxidizing agent, which is why it is so toxic to humans. The formation of photochemical smog can be seen by the following reactions:
NO2(g) -> NO(g)+O(g)
O2(g)+O(g) -> O3(g)
NO2(g)+O2(g) -> NO(g)+O3(g)
In the stratosphere, or the upper atmosphere, the partial pressure of ozone goes to a max of about 10^-5 atm at an altitude of 30 km. Between 95-99% of sunlight in the UV range of 200-300 nm is absorbed by ozone, which we refer to as the ozone layer. The conversion of ultraviolet radiation to thermal energy can be represented by the following equations:
O3(g)+UV radiation -> O2(g)+O(g)
O2(g)+O(g) -> O3(g)+heat
UV radiation -> heat
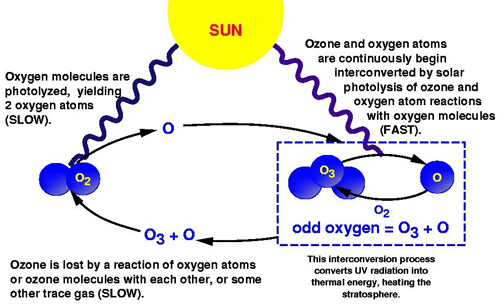
The Chapman Cycle is a process in the stratosphere where there are constant conversions between different oxygen molecules and ozone is continuously being created and destroyed, which can be seen below:
1) An oxygen molecule is photolyzed by solar radiation to create two oxygen atoms:
O2 + hv -> 2O
2) Each oxygen atom reacts with an oxygen molecule to produce ozone:
O2 + O -> O3
3) Then an ozone molecule reacts with an oxygen atom to form two oxygen molecule:
O3 + O -> 2O2
4) Ozone can react with a photon to recycle molecular oxygen:
O3 + hv -> O2 + O
Note: O2 is constantly returning to the atmosphere due to photosynthesis.
CFCs
Chlorofluorocarbons (CFCs) are chemicals usually found in spray aerosols used by industrialized nations. These molecules, which are made up of chlorine, fluorine, and carbon atoms, slowly make their way up into the stratosphere. When the CFCs come into contact with the high energy photons that are found in that region of the atmosphere , they separate into their individual components. Chlorine atoms are especially capable of decomposing ozone molecules because chlorine acts as a catalyst:
O3(g)+Cl(g) -> O2(g)+ClO(g)
ClO(g)+O(g) -> Cl(g)+O2(g)
O3(g)+O(g) -> 2O2(g)
In the troposphere, or the lowest atmosphere, ozone can produce photochemical smog, which contains major chemical pollutants that pose a threat to the environment. It is a powerful oxidizing agent, which is why it is so toxic to humans. The formation of photochemical smog can be seen by the following reactions:
NO2(g) -> NO(g)+O(g)
O2(g)+O(g) -> O3(g)
NO2(g)+O2(g) -> NO(g)+O3(g)
In the stratosphere, or the upper atmosphere, the partial pressure of ozone goes to a max of about 10^-5 atm at an altitude of 30 km. Between 95-99% of sunlight in the UV range of 200-300 nm is absorbed by ozone, which we refer to as the ozone layer. The conversion of ultraviolet radiation to thermal energy can be represented by the following equations:
O3(g)+UV radiation -> O2(g)+O(g)
O2(g)+O(g) -> O3(g)+heat
UV radiation -> heat
The Chapman Cycle is a process in the stratosphere where there are constant conversions between different oxygen molecules and ozone is continuously being created and destroyed, which can be seen below:
1) An oxygen molecule is photolyzed by solar radiation to create two oxygen atoms:
O2 + hv -> 2O
2) Each oxygen atom reacts with an oxygen molecule to produce ozone:
O2 + O -> O3
3) Then an ozone molecule reacts with an oxygen atom to form two oxygen molecule:
O3 + O -> 2O2
4) Ozone can react with a photon to recycle molecular oxygen:
O3 + hv -> O2 + O
Note: O2 is constantly returning to the atmosphere due to photosynthesis.
CFCs
Chlorofluorocarbons (CFCs) are chemicals usually found in spray aerosols used by industrialized nations. These molecules, which are made up of chlorine, fluorine, and carbon atoms, slowly make their way up into the stratosphere. When the CFCs come into contact with the high energy photons that are found in that region of the atmosphere , they separate into their individual components. Chlorine atoms are especially capable of decomposing ozone molecules because chlorine acts as a catalyst:
O3(g)+Cl(g) -> O2(g)+ClO(g)
ClO(g)+O(g) -> Cl(g)+O2(g)
O3(g)+O(g) -> 2O2(g)
Sources:
http://chemwiki.ucdavis.edu/Physical_Chemistry/Kinetics/Case_Studies%3A_Kinetics/Depletion_of_the_Ozone_Layer
http://environment.nationalgeographic.com/environment/global-warming/ozone-depletion-overview/
http://chemwiki.ucdavis.edu/Physical_Chemistry/Kinetics/Case_Studies%3A_Kinetics/Depletion_of_the_Ozone_Layer
http://environment.nationalgeographic.com/environment/global-warming/ozone-depletion-overview/